Welcome to the Aquatic Chemistry Laboratory
Research in the Aquatic Chemistry Laboratory investigates chemical reactions affecting the fate and transport of heavy metals, radionuclides, and other inorganic contaminants in natural and engineered aquatic systems. Specific topics include reactions at the solid-water interface, drinking water treatment, contaminated site remediation, energy byproducts in the environment, biogeochemistry of metals in soils and sediments, and carbon sequestration.
Research Highlights
Recent News

April 2024, Maya won the Outstanding Environmental Engineering Senior Award. Congrats!
April 2024, Maya won the Outstanding Environmental Engineering Senior Award. Congrats!

March 2024, the ACL group went to the ACS Conference in New Orleans.
March 2024, Yao, Yihang, Elmira, Xicheng, Zehua and Dan went to the ACS conference in New Orleans.

March 2024, Xicheng and Dan went to the NAWI Quarterly Review Meeting in Denver.
March 2024, Xicheng and Dan presented at the NAWI Quarterly Review Meeting in Denver.

January 2024, Priya joined ACL. Welcome!
January 2024, Priya joined our group. She will work with Zehua about POU project. Welcome!

November 2023, Elmira and Xicheng passed their thesis proposal defenses. Congrats!
November 2023, Elmira and Xicheng passed thesis proposal defense and became Ph.D. candidates. Congrats!

November 2023, Yao and Dan attended the Water Quality Technology Conference (WQTC) in Dallas.
November 2023, Yao and Dan attended WQTC in Dallas.

November 2023, Yihang attended the Mid-American Environmental Engineering Conference (MAEEC) at the University of Missouri.
November 2023, Yihang gave a presentation at MAEEC at Mizzou. She presented on Effect of Water Chemistry on Selenium Removal.

July 2023, Andy joined ACL for summer research. Welcome!
July 2023, Andy joined our group for summer research. He will work with Yihang.


June 2023, Weiyi Receives Two Best Dissertation Awards at AEESP Conference.
June 2023, our alumnus Weiyi received the 2022 Paul V. Roberts/AEESP Outstanding Doctoral Dissertation Award and one of the 2023 CAPEES-Elsevier Dissertation Awards at the AEESP Conference. Congrats to Weiyi! Please check the link for more information.

March 2023, Yao, Yihang, Elmira, Xicheng and Dan went to ACS conference in Indianapolis.
March 2023, Yao, Yihang, Elmira, Xicheng and Dan went to the ACS conference in Indianapolis. We met our alumnus Yin and his students.

February 2023, Yihang and Dan went to the NAWI Quarterly Review Meeting in Denver.
February 2023, Yihang and Dan presented at the NAWI Quarterly Review Meeting in Denver.

December 2022, Zehua Wang joined our lab, welcome!
December 2022, Zehua joined the ACL as a Ph.D. student. Welcome!

December 2022, Zimeng Wang has been selected as one of four winners of the 2023 James J. Morgan Early Career Award ! Congrats to Zimeng!
Our alumnus Zimeng Wang has been selected as one of four winners of the 2023 James J. Morgan Early Career Award. Congrats to Zimeng!

November 2022, Anushka Mishrra passed her final defense! Congrats!
Anushka passed her final defense. Congratulations, Dr. Mishrra!

November 2022, Yihang Yuan passed her proposal defense! Congrats!
Yihang passed her thesis proposal defense and became a Ph.D. candidate.


November 2022, Jean Brownell and Zehua Wang, first-year PhD students, joined us for rotation. Welcome!
Jean received her B.S. in Chemical Engineering from University of Rochester. She will work with Anushka.
Zehua received his B.S. in Environmental Engineering from Duke University. He will work with Yao.

October 2022, Yao received one of the best presentation awards at the MAEEC. Congrats!
Yao received one of the best presentation awards at the MAEEC. Congrats to Yao!

September 2022, Xueyi Liu, a first-year PhD student joined us for a research rotation. Welcome!
Xueyi received her B.S. in Environmental Science from Nankai University in 2022. She will work with Elmira.

September 2022, Das Bedadeep, a first-year PhD student joined us for a research rotation. Welcome!
Das received his B.Tech and M.Tech in Biotechnology from Indian Institute of Technology Guwahati. He will work with Yihang.

September 2022, Neha passed her final defense. Congrats!
September 2022, Neha passed her final defense. Congratulations, Dr. Sharma!

June 2022, Dan received an AEESP Distinguished Service Award for his leadership of the AEESP Research and Education Conference.
Dan and prof. Jun received distinguished service awards from the Association of Environmental Engineering and Science Professors (AEESP).

June 2022, Bella and Maya join ACL for summer research. Welcome!
Bella and Maya join ACL for 10-week summer research. Bella works with Yao, and Maya works with Yihang.

June 2022, Elmira and Xicheng passed the qualifying exam. Congrats!
Congratulations to Elmira and Xicheng on passing the qualifying exam!

May 2022, Weiyi passed his final defense. Congrats!
May 2022, Weiyi passed his final defense. Congratulations, Dr. Pan!

April 2022, Anushka was selected as a winner of one of the 2022 Graduate Student Paper Awards.
Anushka Mishrra was selected as a winner of one of the 2022 Graduate Student Paper Awards. This is the highest award given to students by the Division of Environmental Chemistry of the American Chemical Society (ACS). She presented her paper in the Fall ACS Meeting in Chicago at the special C. Ellen Gonter Environmental Chemistry Awards Symposium.

April 2022, Weiyi won first place in the Fresh Ideas Student Poster competition. Congrats!
Weiyi won first place in the Fresh Ideas Student Poster competition. As the competition winner he represented Missouri at the AWWA Annual Conference and Exposition in June in San Antonio. Check news for more information.
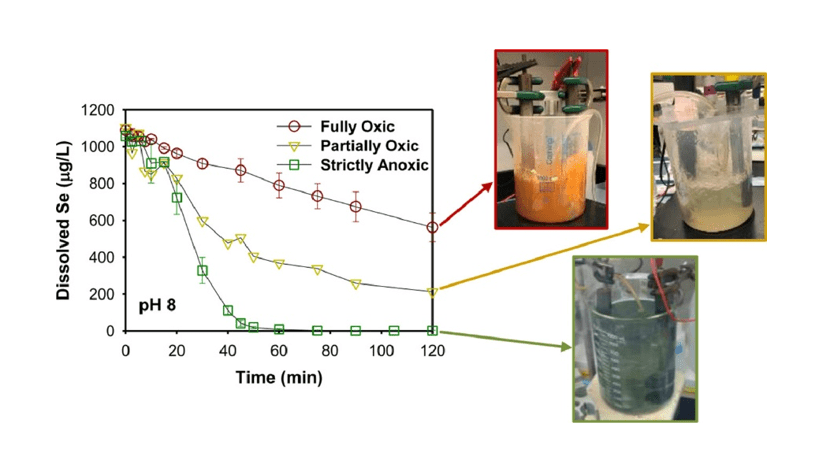
February 2022, Yeunook’s on selenium removal from water was published in Water Research.
Yeunook’s work impact of dissolved oxygen and pH on the removal of selenium from water by iron electrocoagulation got published on Water Research. Congrats!

January 2022, Neha got a Graduate Student Award from ACS Environmental Chemistry Division.
January 2022, Neha got the Graduate Student Award from ACS Environmental Chemistry Division. Congrats!

January 2022, our collaboration with UIUC was published in Water Research.
January 2022, our collaboration with UIUC on the point-of-use filter got accepted by Water Research. Congrats!

December 2021, Weiyi got a Student Travel Award from the ACS Geochemistry Division.
December 2021, Weiyi got the Spring 2022 Student Travel Award from the ACS Geochemistry Division. It will offer registration reimbursement for the ACS National Meeting and an extended time allocation for his oral presentations.

December 2021, Elmira and Xicheng joined our lab, welcome!
December 2021, Elmira and Xicheng joined the ACL as Ph.D. students. They will work on the rare earth elements project.
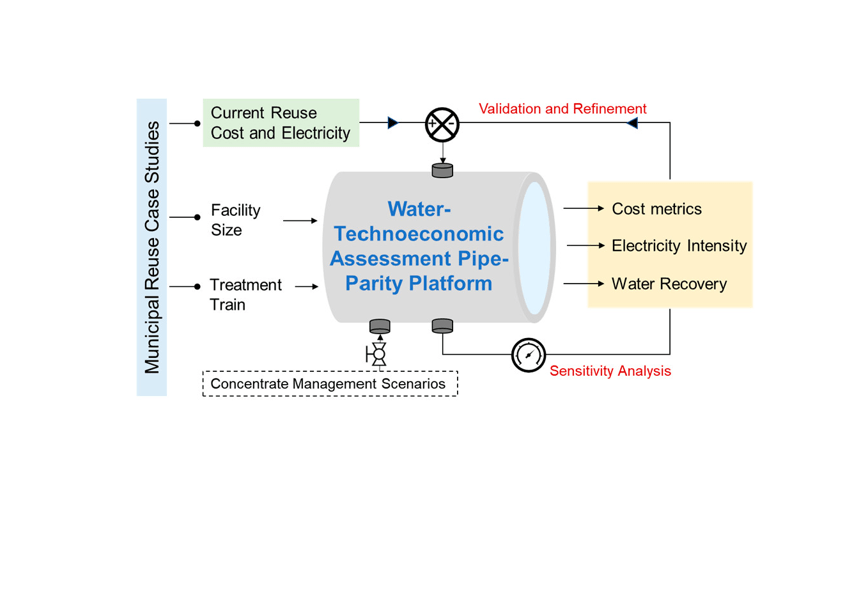
December 2021, Dan and Anushka’s work is accepted by ACS EST Engineering. Congrats!
Dan and Anushka’s work Cost and Energy Metrics for Municipal Water Reuse is accepted by ACS EST Engineering. Please check the attached link for more information.

December 2021, Yao Ma passed his proposal defense! Congrats!
December 2021, Yao passed his proposal defense. Congrats!

Elmira Ramazanova, a first-year PhD student, joined us for a 6-week research rotation. Welcome!
November 2021, Elmira joined our group as a rotation student. She obtained her BEng and MSc in Civil and Environmental Engineering from Nazarbayev University in 2019 and 2021. She will work with Neha.

Hakyung Lee, a first-year PhD student, joined us for a 6-week research rotation. Welcome!
November 2021, Hakyung joined our group as a rotation student. She obtained her BSc from Kyung Hee University and MSc in Environmental Engineering from Seoul National University. She will work with Anushka.

September 2021, Anushka won the 3MT comptetition. Congrats!
September 2021, Anshuka won the MAEEC 3MT competition. Her presentation topic is Lead In Your Drinking Water: Is It Really Safe?

September 2021, WashU successfully held Mid-American Environmental Engineering Conference (MAEEC)!
September 2021, Mid-American Environmental Engineering Conference (MAEEC) was successfully held in Washington University in St. Louis. We had 78 participants, 28 great student presentations, and an inspiring keynote presentation from Dr. Vern Snoeyink.

Xicheng He, a first-year PhD student, joined us for a 6-week research rotation. Welcome!
September 2021, Xicheng joined our group as a rotation student. He obtained his B.S. in Environmental Science from Nankai University in 2021. He will work with Neha.


September 2021, Jeff and Dan won a three-year, $2.25 million award from the DOE. Congrats!!
Jeff and Dan won a three-year, $2.25 million award from the Office of Basic Energy Sciences at the U.S. Department of Energy (DOE). The award supports Catalano’s project “Geochemical Mechanisms Controlling Rare Earth Element and Platinum Group Element Migration and Enrichment during Crustal Aqueous Alteration and Weathering.”

September 2021, Anushka’s work is accepted by Environmental Science & Technology Letters. Congrats!
Anushka’s work Estimating lead concentrations in drinking water after stagnation in lead service lines using water quality data from across the United States is accepted by ES&T Letters. Congrats!

September 2021, Weiyi’s work is accepted by Environmental Science & Technology. Congrats!
Weiyi’s work Effects of Cu(II) and Zn(II) on PbO2 Reductive Dissolution under Drinking Water Conditions: Short Term Inhibition and Long Term Enhancement is accepted by ES&T! This work will be published on the special issue of Environmental Science & Technology that will be a tribute to Dr. James J. Morgan. Congrats!

August 2021, Max joined ACL. Welcome!
Max Trachtenberg is a sophomore at WUSTL studying Environmental Engineering. He joins the ACL in Fall 2021 and works with Anushka.

August 2021, Andrea finished her two-month research at ACL
Andrea Alemán-Reyes finished her two-month research program with Weiyi. She is back at her university and will be a fourth-year student soon.

Dan will serve as conference chair for 2021 AEESP
The 2021 AEESP Research and Education Conference will be hosted by Washington University in St. Louis together with 3 other universities! Dan will serve as the Conference Chair.

The 2021 AEESP Virtual “Appetizer” registration is open now!
The registration for the 2021 AEESP Conference “Appetizer” is open now. This event includes a keynote, select workshops, a “meet the candidate” session, and the AEESP president’s address and awards.

June 2021, Anshuman’s work is accepted by Environmental Science & Technology. Congrats!
Anshuman’s work of intercomparison and refinement of surface complexation models for U(VI) adsorption onto goethite based on a metadata analysis is accepted by ES&T! This work is a collaboration with Dr. Zimeng Wang. Congrats!

June 2021, Anushka is awarded the 2021 Abel Wolman Fellowship. Congrats!
Anushka is awarded the 2021 Abel Wolman Fellowship by the American Water Works Association. Congratulations! Check the news from McKelvey School of Engineering

June 2021, Yihang is officially a member of ACL. Welcome!
Welcome and congratulations, Yihang! She passed the qualifying exam and joined the ACL. She was working with Weiyi for her rotation. Now, she is working with Anushka.

June 2021, Andrea joined the ACL for a two-month summer research program.
Andrea Alemán-Reyes is a third-year undergraduate student at the University of Puerto Rico. She will spend her summer here doing exciting research. Currently, she is working with Weiyi.

May 2021, Dan hosted a party to celebrate Anshuman and Elizabeth’s graduations.
Dan hosted a party at his house to celebrate Anshuman and Elizabeth’s graduations. People in the picture are (left to right): Yao, Weiyi, Neha, Anshuman, Elizabeth, Dan, and Anushka. Congratulations to Anshuman and Elizabeth!

April 2021, ACL members attended the 2021 ACS Spring Conference.
Anshuman, Kyle, Neha, and Weiyi gave presentations about their works at the 2021 ACS Spring Conference.

Anshuman will join the University of Notre Dame as a postdoctoral researcher.
Anshuman will join Dr. Amy Hixon‘s group at the University of Notre Dame as a post-doc in June 2021. Congratulations, Dr. Satpathy!

April 2021, Kun-Pu, Jerry, and Yihang finished their research rotations at ACL.
Kun-Pu, Jerry, and Yihang have finished their 6-week research rotations with Anushka, Neha and Weiyi. All of them did a great job in the past 6 weeks. Good luck with their qualifying exams!

Kun-Pu Ho, a first-year PhD student, joined us for a 6-week research rotation. Welcome!
Kun-Pu Ho, a first-year PhD student, joined us for a 6-week research rotation. Welcome!

Zhenqin (Jerry) Wang, a first-year PhD student, joined us for a 6-week research rotation. Welcome!
March 2021, Jerry joined our group as a rotation student. He obtained his B.S. in Energy Chemical Engineering from Changzhou University in 2018 and M.S.E in Chemical & Biomolecular Engineering from Johns Hopkins University in 2020. He will work with Neha.

Yihang Yuan, a first-year PhD student, joined us for a 6-week research rotation. Welcome!
March 2021, Yihang joined our group as a rotation student. She obtained her bachelor’s degree from the Hefei University of Technology and master’s degree from Johns Hopkins University. She will work with Weiyi.

March 2021, Ying and Zixuan finish their research rotations at ACL
Ying and Zixuan have finished their 6-week research rotations with Anushka and Neha. Both of them did a great job in the past 6 weeks. Good luck with their next rotations!

March 2021, Anushka receives award from ACS envr
Anushka has been selected as a winner of the 2021 Graduate Student Award in Environmental Chemistry by the Division of Environmental Chemistry of the ACS.

February 2021, Weiyi’s paper is published on Environmental Science&Technology Letters.
Weiyi and Elizabeth found that Pb phosphate particles can be a challenge for point-of-use filters to remove.

February 2021, Anshuman passed his thesis defense.
February 22th, Anshuman passed his thesis defense. Congratulations, Dr. Satpathy!

We shared our findings of Pb in tap water on AWWA MO Show Me Magazine
January 2021, our lab has been assessing the effectiveness of corrosion control treatment techniques and enabling new tools for understanding Pb exposure from tap water. Read the Show Me Magazine article

Ying Wang, a first-year PhD student, joined us for a 6-week research rotation. Welcome!
January 2021, Ying joined our group as a rotation student. She obtained her B.Eng. from the South China University of Technology in 2017 and M.S. from Tongji University in 2020. She is currently working with Anushka.

Zixuan Wang, a first-year PhD student, joined us for a 6-week research rotation. Welcome!
January 2021, Zixuan joined our group as a rotation student. He obtained his B.S. from Xi’an Jiaotong University in 2018 and M.S. from Virginia Polytechnic Institute and State University in 2020. He is currently working with Neha.

Researchers devise new method to get lead out of filters, better measure amount in tap water
October 2020, our research is highlighted by the McKelvey School of Engineering!

September 2020, Anushka’s paper is published on Water Research
The effect of sodium silicate addition on lead release from lead service lines (LSLs) was investigated using laboratory-based pipe loop experiments with LSLs harvested from a water utility that has one of the Great Lakes as its source water.

July 2020, Weiyi’s paper is published on Environmental Science: Water Research & Technology
Weiyi and Elizabeth developed an extraction method that achieves complete recovery of accumulated lead, which will enable its application in assessing potential lead exposure from drinking water in real homes.

July 2020, Weiyi’s paper is published on Environmental Science: Water Research & Technology
Rhamnolipid-coated iron oxide nanoparticles were demonstrated to be able to adsorb the environmentally significant contaminant U(VI), and the effects of water chemistry on adsorption were successfully modeled within a reaction-based surface complexation modeling framework.

May 2020, Guiwei’s work is highighted on the Department’s website
Aluminum may affect lead levels in drinking water. Researchers find aluminum in water could affect lead’s solubility — in certain cases.

May 2020, Yao has passed the qualifying exam

April 2020, Dan receives outstanding teaching award from AEESP
Dan has been awarded the 2020 Association of Environmental Engineering and Science Professors’ (AEESP) Award for Outstanding Teaching in Environmental Engineering & Science.
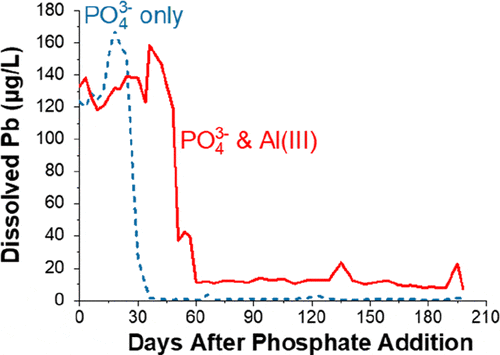
April 2020, Guiwei’s paper is highlighted as ACS Editors’ Choice by Environmental Science & Technology
How will the input of dissolved aluminum to distributed drinking water affect lead release from corrosion scales? Check this study.


March 2020, Yuenook’s paper is published on Water Research
We built pipe loop reactors with lead pipes harvested from Providence, RI, and we operated them with high pH and low alkalinity water of a composition similar to that in Providence.

March 2020, Weiyi receives award from ACS envr
Weiyi has been selected as a winner of 2020 Graduate Student Award in Environmental Chemistry by the Division of Environmental Chemistry of the ACS.

January 2020, Keeping lead out of drinking water when switching disinfectants
Our resarch is hightlighted by the McKelvey School of Engineering! Check this.

Octber 2019, Anushka received the best presentation award in MAEEC
We built pipe loop reactors with lead pipes harvested from Providence, RI, and we operated them with high pH and low alkalinity water of a composition similar to that in Providence.





